SPOTLIGHT
Letter from Dr. Stephen I. Katz: Addressing Scientific Opportunities Through Collaboration and Coordination
Dear Colleagues,
Public health advances, and the findings on which these improvements are based, require collaboration. These interactions vary in formality. Most occur spontaneously while investigators engage in their daily activities. Likewise at the National Institutes of Health (NIH), NIAMS staff are constantly sharing ideas with colleagues across the 27 Institutes and Centers to the benefit of the scientific community. When the NIAMS co-funds a grant, participates in another Institute’s funding opportunity announcement or co-sponsors a workshop, the genesis of our involvement can often be linked to one or more of these informal interactions.
Image: Stephen I. Katz, M.D., Ph.D.
NEWS
NIH Provides Updated Information, Tools and Resources Regarding Clinical Trial Requirements for Grants and Contracts
The NIH is launching a series of initiatives in 2017–2018 to enhance the accountability and transparency of clinical research. These initiatives target key points along the whole clinical trial lifecycle from concept to results reporting. Investigator resources—including training resources and frequently asked questions—can be found on the NIH Office of Extramural Research Clinical Trials website.
NIAMS Issues STAR Awards To Enhance Support for Early-Established Investigators
The NIAMS has awarded Supplements to Advance Research (STAR) program funding to four early-established investigators who have renewed their first NIAMS-funded R01 grants. The supplement enables these scientists to pursue innovative and high-risk research within the broader scope of a current NIAMS-funded, peer-reviewed research project.

Image: (From left to right) Karen Costenbader, M.D., M.P.H.; Ivo Kalajzic, M.D., Ph.D.; Chuanju Liu, Ph.D.; and Rui Yi, Ph.D.
Photo credits: Harvard Medical School, University of Connecticut Health Center, New York University Langone Medical Center, University of Colorado Boulder.
NIAMS Highlights Collaborative Research at Lupus Summit
NIAMS Deputy Director Robert H. Carter, M.D., took part in a National Policy Summit hosted by the Lupus Foundation of America in downtown Washington, DC, on June 26, 2017. Dr. Carter represented the NIAMS and the NIH in two panels during the daylong event, which brought together lupus clinician and patient advocacy communities to discuss promising future research directions to improve the lives of people with lupus.
Image: NIAMS Deputy Director Robert H. Carter, M.D., (third from left) speaks on the importance of lupus research.
Photo credit: Colleen Dundas, M.P.H., NIAMS.
NIH Study Uncovers Specialized Mouse Neurons That Play a Unique Role in Pain: Previously Unknown Category of Neuron Responds to Pulling of a Single Hair
Researchers from the NIH have identified a class of sensory neurons (nerve cells that electrically send and receive messages between the body and brain) that can be activated by stimuli as precise as the pulling of a single hair. Understanding basic mechanisms underlying these different types of responses will be an important step toward the rational design of new approaches to pain therapy.
Photo credit: Jeremy and Nichole Swan.
Stay Up to Date on the All of Us Research Program
The mission of the All of Us Research Program is to accelerate health research and medical breakthroughs, enabling individualized prevention, treatment and care for all of us. See the latest news, and subscribe to receive email updates if you’re interested.
Trans-NIH Strategic Plan for Research on Women's Health: Stakeholder Feedback Requested
The NIH Office of Research on Women’s Health is updating the Trans-NIH Strategic Plan for Women’s Health. A Request for Information solicits input from basic, clinical and translational scientists as well as advocacy and patient communities on topics under consideration for the next strategic plan. Responses are due November 10, 2017.
NCATS Launches Toolkit for Patient-Focused Therapy Development
On September 8, 2017, the NIH’s National Center for Advancing Translational Sciences (NCATS) launched its Toolkit for Patient-Focused Therapy Development online resource portal. Developed in collaboration with patients and rare disease advocates, the toolkit is a centralized online portal for resources and tools that will cover the broad therapy development landscape, including how to:
- Establish a patient registry.
- Drive patient-focused discovery and preclinical research and development.
- Work with the NIH and the U.S. Food and Drug Administration (FDA).
- Conduct post-market surveillance.
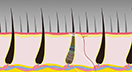
Missing Strands? Dealing With Hair Loss
Hair loss can happen to anyone, and whether hair might eventually grow back depends on the cause. This feature article discusses types of hair loss—with special attention to alopecia areata, an autoimmune disease. NIAMS-funded research has shown some promising results for treating alopecia areata with a class of drugs called Janus kinase (JAK) inhibitors.
Mindfulness-Based Stress Reduction, Cognitive-Behavioral Therapy Shown To Be Cost Effective for Chronic Low Back Pain
Group sessions of either mindfulness-based stress reduction or cognitive-behavioral therapy provide cost-effective treatment for chronic low back pain, according to research supported by the National Center for Complementary and Integrative Health (NCCIH) and published in the journal Spine.
Photo credit: ThinkStock.
Kids in Research: Your Child Can Help Find Cures at the NIH Clinical Center
Clinical research relies on volunteers to advance medicine. The NIH offers a website where parents can learn about how children can participate in clinical trials as a way to help researchers find future cures for today’s illnesses. This fact sheet (PDF – 303KB) has more information.
FDA: Ticks and Lyme Disease: Symptoms, Treatment and Prevention
This Consumer Update from the FDA includes a video, “Lyme Disease: Diagnosis and Treatment for People and Pets,” statistics about increases in tick-borne disease cases in the United States and information on symptoms, treatment, prevention and how to remove a tick safely.
RESOURCES
Spotlight on Scientific Imagery: Muscle-Specific Stem Cells on Mouse Muscle Fibers
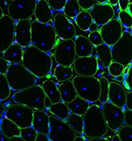
This cross-section of regenerated muscle shows muscle stem cells (red) in their niche along the muscle fibers (green). (View the full image to see all the details.) The blue dots are DNA in the nuclei of the fibers. Researchers have found that injecting the molecule prostaglandin E2 into muscles after injury induces muscle stem cell division and accelerates regeneration. Prostaglandin E2 is an inflammatory molecule released in response to muscle injury or rigorous exercise.
Photo credit: Helen M. Blau, Ph.D., Andrew T.V. Ho, Ph.D., and Adelaida R. Palla, Ph.D., Stanford University School of Medicine.
EVENTS
September NIAMS Advisory Council Meeting
The September 6 NIAMS Advisory Council archived videocast is now available in the Past Events section of the NIH Videocasting website.

NCATS Workshop on Translational Challenges of Induced Pluripotent Stem Cells
September 26, 2017
8:15 a.m. to 5 p.m.
National Library of Medicine, Building 38A, NIH Campus, Bethesda, Maryland
Register here.
A meeting agenda is available.
FDA Inaugural Meeting of New Patient Engagement Advisory Committee
October 11, 2017, from 1 p.m. to 5 p.m., and October 12, 2017, from 8 a.m. to 5 p.m.
Hilton Washington DC North, Gaithersburg, Maryland
Discussion will include challenges in clinical trial design, conduct and reporting, as identified by patients.
Additional information is available here.
Facts and Uncertainties: Science and the Practice of Medicine
October 17, 2017
2 p.m. to 3:30 p.m.
Building 10, Lipsett Auditorium, NIH Campus, Bethesda, Maryland
Speaker: Josephine Briggs, M.D., Director, NCCIH
Videocast is available.
Sex as a Biological Variable Workshop
October 26–27, 2017
NIH Campus, Bethesda, Maryland
Cost: Free
Details and registration information available here.
NIH Science Lectures and Events Available via Internet
The NIH hosts a number of science seminars and events that are available online through real-time streaming video (videocast). The NIH calendar notes these videocast events with a video icon ![]() .
.